Co-authored with Albert Tenuta, Field Crop Pathologist, OMAFRA
OMAFRA field crop specialists in collaboration with Grain Farmers of Ontario (GFO) and members of the Ontario Agri-Business Association (OABA) have completed the annual Ontario corn ear mould and DON mycotoxin survey. Corn ear moulds such as Gibberella and their corresponding mycotoxins occur every year in Ontario. These mycotoxins, particularly deoxynivalenol (DON, also referred to as vomitoxin) are produced primarily by Gibberella/Fusarium ear moulds and can be disruptive when fed to livestock, especially hogs. The 2020 survey found 89% of samples tested low (<2.00 parts per million (ppm)) for DON which is in line with long term survey averages.
Methods:
From September 23 to October 2, 2020, 245 ear corn samples were collected from across the province. Five consecutive ears were collected from four random locations throughout a field and placed into high temperature driers (80°C) as soon as possible after collection. Pictures were taken to document moulds (Fig. 1) as well as insect and bird feeding damage. Dry ears were shelled and coarse ground and mixed for sub-sampling consistency. Sub samples were collected and finely ground for DON analysis by quantitative ELISA analysis at the University of Guelph Ridgetown Campus mycotoxin lab.

Figure 1. Corn ear moulds which can be observed in Ontario.
Results:
Table 1. Deoxynivalenol (DON) results from the 2020 Ontario grain corn ear mould and DON survey.
| DON Concentration parts per million (ppm) | All Samples |
| Total Samples | 245 |
| < 0.50 ppm | 58% |
| 0.50 to <2.00 ppm | 31% |
| 2.00 to <5.00 ppm | 10% |
| 5.00 ppm and greater | 0% |
Results for the 2020 survey are presented in Table 1. Visual mould and DON concentrations were in line with long term survey averages (Table 2). While ear moulds were certainly evident in some samples, many displayed no symptoms, and only one sample tested over 5 ppm. While insect feeding damage was lower than many past years, feeding damage was often associated with greater visual ear moulds, and several samples with higher DON concentrations were those that had been damaged. Other mouldy samples with no ear damage were observed where weather and hybrid susceptibility were likely factors.
Table 2. DON results from the past ten Ontario grain corn ear mould and DON surveys.
| DON Concentration (ppm) | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | 2019 | 2020 |
| < 0.50 | 47% | 84% | 83% | 66% | 75% | 48% | 69% | 33% | 84% | 58% |
| 0.50 to <2.00 | 28% | 12% | 15% | 26% | 20% | 26% | 17% | 27% | 12% | 31% |
| 2.00 to <5.00 | 17% | 4% | 1% | 6% | 5% | 18% | 8% | 15% | 4% | 10% |
| 5.00 and greater | 7% | 0% | 0% | 2% | 0% | 8% | 6% | 25% | 0% | 0% |
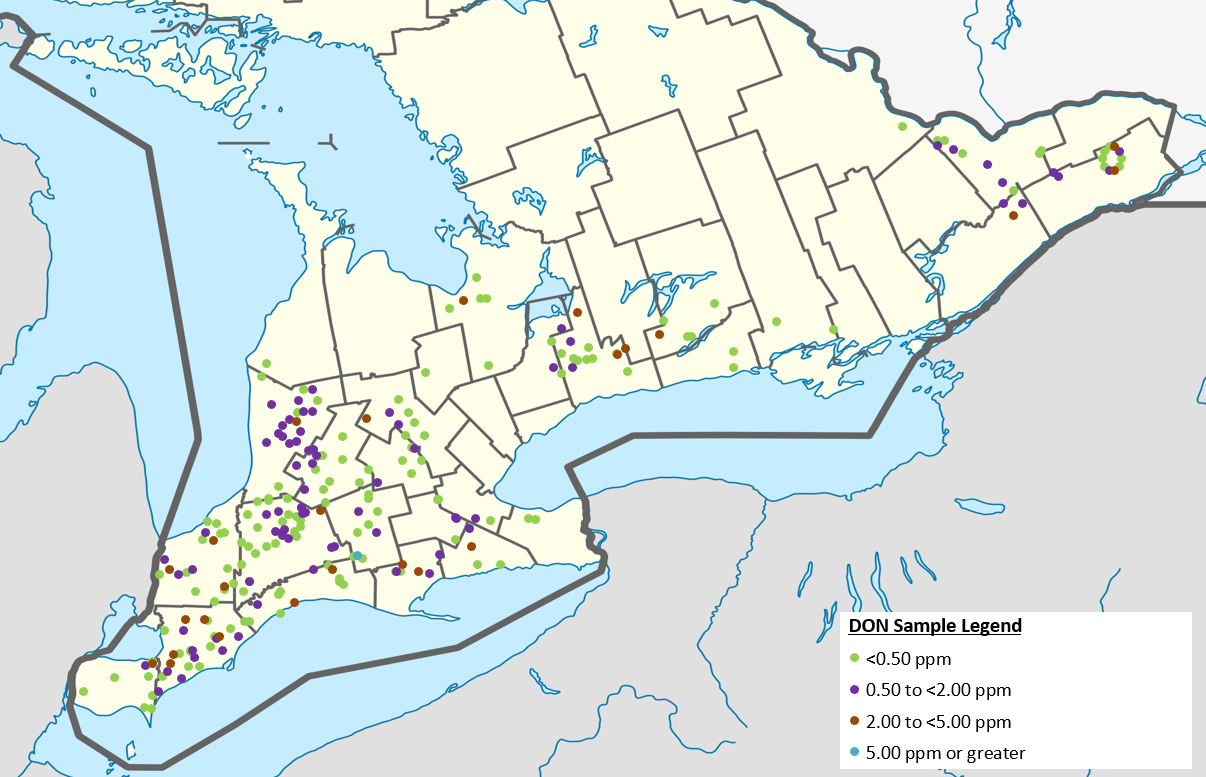
Figure 2. Corn ear mould and deoxynivalenol (DON) survey sampling locations and results in 2020.
While 89% of 2020 samples tested below 2 ppm, growers should still remain vigilant and understand management options in fields with higher DON concentrations. Ear moulds and mycotoxins occur every year in Ontario and fields need to be assessed individually. Distribution of samples and their corresponding DON levels are presented in Figure 2.
Growing Season
The growing season remained relatively dry across the province from April through to mid-July. As corn started tasseling mid-July, regular rainfall events occurred for many areas of the province which were good for corn yields but also could provide favourable environmental conditions ( moisture and humidity) in corn canopies for ear mould establishment at fresh silk stage. Frequent rains persisted for several weeks following tasselling. This along with high temperatures may have contributed to ear moulds establishing in fields, particularly those with hybrids known to be more susceptible to ear moulds, and those with ear feeding damage. Drier conditions through later August and September likely limited ear mould progression in fields where infection was established. During the survey, corn maturity ranged from half-milk to black layer.
Feeding Damage:
Ear feeding by pests, particularly western bean cutworm (WBC), corn earworm (CEW) or birds can open husks, damage kernels and present opportunities for greater ear mould infection and DON. Feeding damage appeared lower than normal in 2020 survey samples, likely a result of most corn tasseling prior to peak WBC moth flights. Feeding from WBC or CEW was present in some samples however and appeared to elevate ear mould and DON where present.
How to Sample Corn Loads:
Several steps can impact the accuracy of DON sampling:
The importance of collecting representative samples cannot be emphasized enough. Significant variability in mycotoxin testing can come from poor sampling. While sampling from the top of a storage bin, truck or combine may be convenient, mycotoxins are rarely distributed evenly in grain loads. If the sample comes from a bin, truck, wagon or other stationary corn load, a sample probe is recommended. Ten probe samples are recommended, while 5 is considered a minimum. For moving grain streams, use a diverter or randomly collect cupfuls of grain. Group and mix samples into one bulked sample.
One of the most significant sources of variability is from subsampling the bulked sample for analysis. Because individual kernels can be highly variable and still coarse in size relative to the subsample size, the whole bulked sample should be ground (at least coarsely) and mixed to improve ability to collect a representative subsample of the bulked sample.
A GFO, OABA, OMAFRA supported University of Guelph Ridgetown Campus project lead by Dr. Art Schaafsma has resulted in the development of a high capacity sample grinder which based on laboratory trials shows considerable promise in addressing the sample variability associated with DON testing. A pilot project utilizing these grinders in commercial operations is being planned for this fall.
Last, samples must be processed quickly – ship or deliver fresh sample promptly. The longer the sample sits around the greater potential for inaccurate results.
Going Forward:
This survey does not capture all regions of the province and results can vary from field to field depending on local weather, hybrid, planting date, insect feeding, rotation, residue levels, fungicide practices and moisture. Results may not capture what is occurring in your field, therefore monitoring is always recommended. Timely harvest is important. Leaving diseased grain in the field allows ear rot fungi to continue growing, which increases risks of mouldy grain and mycotoxin contamination. Most ear rot fungi continue to grow (and potentially produce mycotoxins) until grain moisture is below 15%. In high risk or severely infected fields, growers should consider harvesting at higher moisture and drying below 15%.
If a field contains significant ear moulds (10% or more ears with mould symptoms) collect a representative sample prior to harvest and test for mycotoxins before storing or feeding to livestock. A lab test is the only reliable way of determining mycotoxin levels.
If possible, segregate contaminated corn separately.
When ear rots are present, the following harvest, storage and feeding precautions are advisable (adapted from OMAFRA Pub 811, Agronomy Guide for Field Crops):
- Harvest and dry as quickly as possible, especially susceptible hybrids. Mould and mycotoxin development ceases with grain moistures below 15%.
- If insect or bird damage is confined to certain areas, harvest and handle damaged rows separately.
- Adjust harvest equipment to minimize grain damage and to remove insect damaged, infected or smaller end kernels. Clean corn thoroughly to remove cob, small kernels and red corn dust (red dog) which may be infected.
- Clean bins before storing new grain and cool grain after drying. If possible, segregate corn by DON content to help match end use.
- Check stored grain often for temperature, wet spots, insects and mould. For grain with elevated mould content, market as soon as possible. Avoid long term storage.
- Exercise caution handling or feeding mouldy corn to livestock, especially hog breeding herds and weanling pigs. Pink or reddish moulds are particularly harmful. Test suspect samples for toxins. Work with a nutritionist to manage DON levels in feed.
Preventing ear rots and mould can be difficult since weather is critical to development. A few things to consider for 2021. Hybrid selection is important, and while tolerant hybrids are available, none have complete resistance. Growers are encouraged to discuss ear mould tolerance with their seed supplier. The Ontario Corn Committee has initiated inoculated DON trials for evaluating differences in DON susceptibility across Ontario hybrids. After environmental issues in 2019, a revised protocol has been established in 2020 and results are expected to be released this fall. Crop rotation may help reduce ear rots, while certain foliar fungicides are also registered for suppressing ear rots. Cultural practices such as tillage are shown to have limited success in preventing ear and kernel rots.
Agricorp customers with mould issues are encouraged to immediately contact Agricorp at 1-888-247-4999 and report damage.
Additional Resources:
For additional information, see the Crop Protection Network’s “Corn Management Disease Series – Ear Rots” publications at CropProtectionNetwork.org
Crop Protection Network Publications:
CPN-2002 – Corn – Mycotoxin FAQs
CPN-2003 – Corn – Grain Sampling and Mycotoxin Testing
CPN-2004 – Corn – Storing Mycotoxin-Affected Grain
Acknowledgements:
Thanks to Grain Farmers of Ontario, DON mycotoxin working group, University of Guelph Ridgetown Campus mycotoxin lab and OMAFRA plant health fund for support of the survey as well as Ontario Agri Business Association (OABA) and its members for their interest and participation. Sincere thanks to those who assist in co-ordination and collection of samples: Agris Co-operative, Anderson Agronomy Services, Belmont Farm Supply, Benjamins Agronomy Services, Campbellford Farm Supply, Clark Agri Service, FS Partners, Hoegy’s Farm Supply, Holmes Agro, Lakeside Grain & Feed, Lockie Farms Grain Elevator, MacEwen Agricentre, Maizex Seeds, Parrish and Heimbecker, P.T. Sullivan Agro, St. Lawrence Grain & Farm Supply, Sylvite, The Andersons, Wellburn Agromart and the many producers and OMAFRA staff also involved. This co-operation is critical for covering as much of the corn growing areas of the province as possible in a short period of time.
Comments are closed.